Endotoxin: study, detection and elimination
What is endotoxin?
Endotoxin is used to describe a class of lipopolysaccharides (LPS) which are an integral part of the outer cell membrane of Gram-negative bacteria. Endotoxins are the structure component of the bacteria cell membrane and responsible for the stability and basic function. By firmly anchored in the cell membrane, endotoxin also participates in the interaction of the bacterial cell with its environment and possible hosts. Endotoxins are heat-resistance macromolecular, the commonly used autoclave sterilization process at 121℃ for 30min does not destroy their toxicity. Endotoxin was termed by R.Pfeiffer (1858–1945) back in the 1890s, which was used to distinguish with exotoxins. Exotoxins are a type of toxin that is generated and released actively by bacteria into the surrounding environment [1, 2].
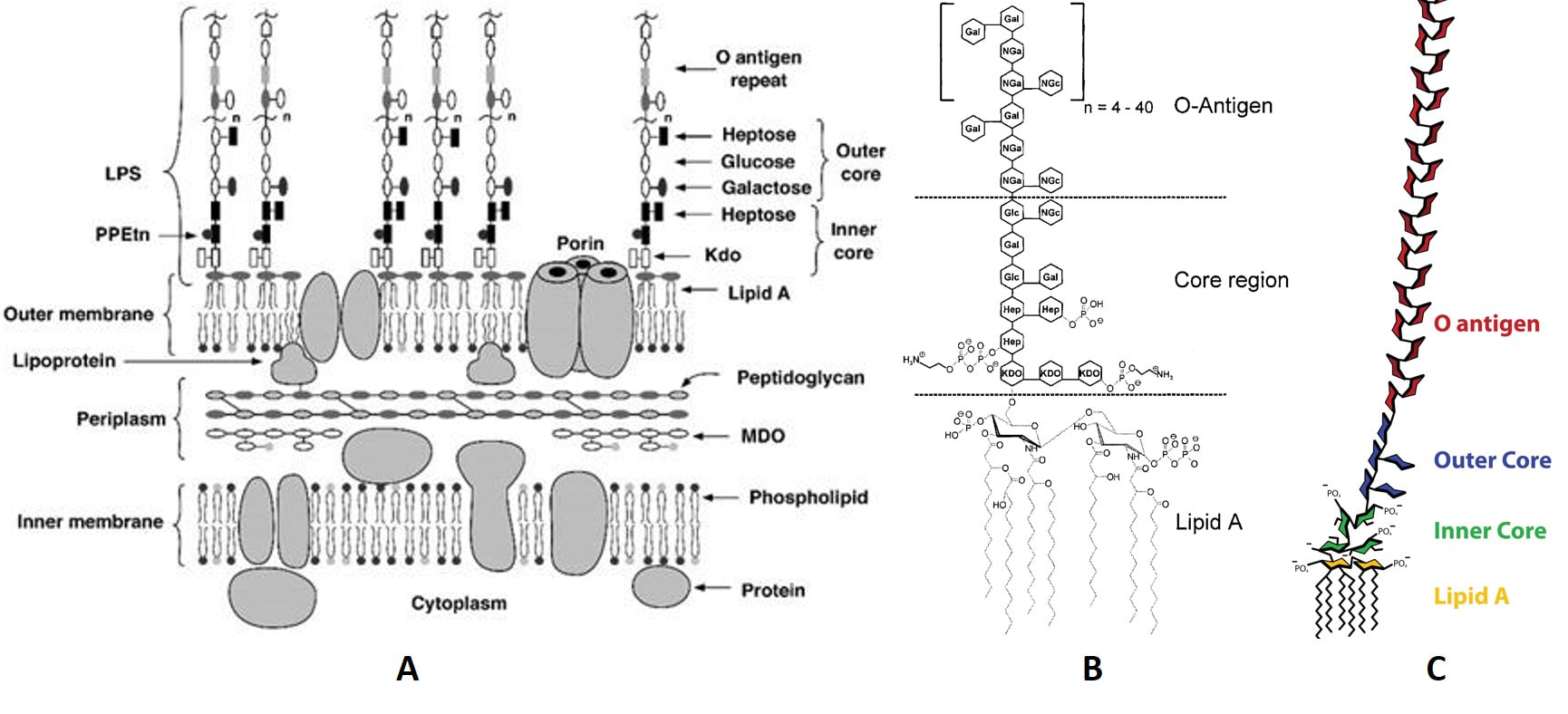
Figure 1. The molecular models of endotoxin and its affiliated structure.
A: The inner and outer membranes of E. coli K-12 in molecular model [6]. Geometric form:
▭ and 0 represent sugar residues;
○ represent polar head groups of various lipids.
Abbreviation: PPEtn (ethanolamine pyrophosphate); LPS (lipopolysaccharide); Kdo (2-keto-3-deoxyoctonic acid).
B: Chemical structure of endotoxin from E. coli O111:B4 according to Ohno and Morrison 1989 [7].
(Hep) L-glycerol-D-manno-heptose; (Gal) galactose; (Glc) glucose; (KDO) 2-keto-3-deoxyoctonic acid; (NGa) N-acetyl-galactosamine; (NGc) N-acetyl-glucosamine.
C: Structure of bacterial lipopolysaccharides (Source: https://en.wikipedia.org/wiki/Lipopolysaccharide, accessed on 21 Dec 2021).
A: The inner and outer membranes of E. coli K-12 in molecular model [6]. Geometric form:
▭ and 0 represent sugar residues;
○ represent polar head groups of various lipids.
Abbreviation: PPEtn (ethanolamine pyrophosphate); LPS (lipopolysaccharide); Kdo (2-keto-3-deoxyoctonic acid).
B: Chemical structure of endotoxin from E. coli O111:B4 according to Ohno and Morrison 1989 [7].
(Hep) L-glycerol-D-manno-heptose; (Gal) galactose; (Glc) glucose; (KDO) 2-keto-3-deoxyoctonic acid; (NGa) N-acetyl-galactosamine; (NGc) N-acetyl-glucosamine.
C: Structure of bacterial lipopolysaccharides (Source: https://en.wikipedia.org/wiki/Lipopolysaccharide, accessed on 21 Dec 2021).
Generally, all of the endotoxins exhibit similar structure with a polar heteropolysaccharide chain, covalently bond to a non-polar lipid moiety (lipid A), as graphically shown in Fig. 1. The endotoxin is anchored in the outer bacterial membrane through lipid A. Being composed of a core oligosaccharide (core region) and a surface antigen (O-antigen), the heteropolysaccharide is exposed to the environment. A series of repeating oligosaccharide units (each of 3–8 monosaccharides) forms a chain named O-antigen. The oligosaccharide units are the determiner for the serological identification of bacteria and they are strain-specific. The O-antigen is not presented on genetically modified strains such as E.coli K12. However, the lacking of O-antigen affects neither the viability nor the biological potency of the endotoxin. The core oligosaccharide has a conserved structure; among different bacteria species the structure shows great similarity. Lipid A is the most conserved component, showing very close structural relationship among different bacterial genera. It is important to be aware of the core region near by Lipid A and Lipid A itself are partially phosphorylated, therefore the endotoxins exhibit a net negative charge (pKa=1.3). The molecular weight of an endotoxin monomer may vary between 10 and 20 kDa depends on the variability of the oligosaccharide chain, as shown in Fig. 1. Extreme cases show that even 2.5kDa (O-antigen-deficient) and 70kDa (very long O-antigen) are found. [8]
What is the problem if endotoxin appears in my experiment?
In living organism, endotoxin does not directly interact against cells or organs of the host, but through stimulation of the immune system, especially through monocytes and macrophages. After being stimulated, these cells release several types of mediators, such as tumor necrosis factor, several interleukins, etc., and free radicals [3, 4], having potent biological activity and being responsible for the adverse effects seen upon endotoxin exposure. These include: affecting structure and function of organs and cells, changing metabolic functions, raising body temperature, triggering the coagulation cascade, modifying hemodynamics, and causing septic shock [5]. These reactions lead to the host organism to suffer from endotoxin shock, tissue injury, and even death.
In cell culture, Endotoxin affects cell bioactivity, which lead to growth rate change and/or function alternate. These effects appear both in vitro and in vivo cell culture. In vitro, plenty of studies show that endotoxin causes a variety of problems for cell culture research. Among the effects documented were the stimulation of leukocyte cultures to produce tissue factors, the induced production of IL-6 in equine macrophages, and the inhibition of murine erythroid colony formation by very low levels (less than 1 ng/mL) of endotoxin. In vivo, endotoxins elicit an inflammatory response in animal studies.[25]
In cell culture, Endotoxin affects cell bioactivity, which lead to growth rate change and/or function alternate. These effects appear both in vitro and in vivo cell culture. In vitro, plenty of studies show that endotoxin causes a variety of problems for cell culture research. Among the effects documented were the stimulation of leukocyte cultures to produce tissue factors, the induced production of IL-6 in equine macrophages, and the inhibition of murine erythroid colony formation by very low levels (less than 1 ng/mL) of endotoxin. In vivo, endotoxins elicit an inflammatory response in animal studies.[25]
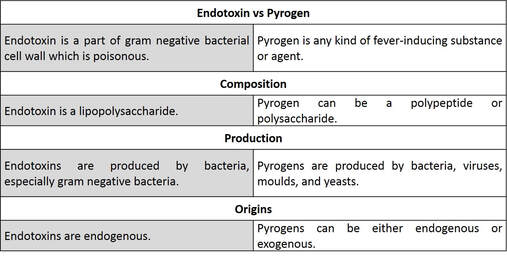
What are the differences between endotoxin and pyrogen?
These 2 terms are often put together and cause confusing. The form hereunder helps to explain the difference.
How do I convert EU/mL to ng/mL of endotoxin?
The most commonly used unit for endotoxin is Endotoxin Units (EU), which was initially defined by the US FDA in order to quantify the endotoxin. Rather than unit of weight, EU is a measure of the activity of endotoxin, which is correlate with biology response. The potency of endotoxin depends on a variety of factors: polysaccharide chain length, aggregation, solubility in biological fluids, bacterial source, associated substances, etc. Expressing endotoxin concentrations in EU addresses the issue of different potencies of different endotoxins.
In order to have better understanding and fast calculation, you can convert the endotoxin concentration by this equation: 10 EU/mL = 1.0 ng/mL.
In order to have better understanding and fast calculation, you can convert the endotoxin concentration by this equation: 10 EU/mL = 1.0 ng/mL.
What are the endotoxin detection methods and what are their principals
Rabbit pyrogen test
The rabbit pyrogen test [9] was the first method approved by US Food and Drug Administration for LPS detection. It was developed in the 1920s on the basis of the rise in temperature of rabbits after an intravenous injection of a test solution. The rabbit pyrogen test has obvious defects related to cost and time period, so it has been gradually replaced by the Limulus Amoebocyte Lysate (LAL) test in practical use. Nowadays, the rabbit pyrogen test is applied only as supplement of the LAL test to analyze pyrogenic materials in the earlier development phase of parenteral devices.
The rabbit pyrogen test [9] was the first method approved by US Food and Drug Administration for LPS detection. It was developed in the 1920s on the basis of the rise in temperature of rabbits after an intravenous injection of a test solution. The rabbit pyrogen test has obvious defects related to cost and time period, so it has been gradually replaced by the Limulus Amoebocyte Lysate (LAL) test in practical use. Nowadays, the rabbit pyrogen test is applied only as supplement of the LAL test to analyze pyrogenic materials in the earlier development phase of parenteral devices.
Limulus amebocyte lysate (LAL) test
The Limulus Amebocyte Lysate (LAL) test was discovered in the 1960s by Levin and Bang.[19] It uses the blood extract of horseshoe crabs, Limulus polyphemus and is mostly known as the indirect animal test. This method can be divided into three basic techniques, the gel-clot, turbidimetric and chromogenic techniques.
Although the LAL test has been accepted as a reliable method for measuring pyrogenicity, it is only specific for the cell wall component of Gram-negative bacteria. Thus, it is not applicable for samples containing a large amount of protein.[11] In the case of endotoxin contamination in veterinary vaccines, the LAL test is not capable for quantifying in vivo endotoxin activities satisfactorily.[12] LAL test is not applicable on samples containing free metal ions as these metal ions can alter the measurement sensitivity, thus reducing the accuracy of the test result. Several types of modification to the method has been employed to reduce the influence of ion. Briefly, the modification involved reconstructing the sample so that the composition of all metal ions in each sample was analogous. A minimum sample dilution (MSD) of 1000-fold was introduced to reduce further the inhibition/enhancement effect of metal ions on the LAL activity without compromising endotoxin sensitivity. 5 mM EDTA was added into each sample to reduce the endotoxin–metal ion interaction. Besides the issues on LAL sensitivity and specificity, the horseshoe crab population is also under threat due to over-fishing for its blood.[13]
The Limulus Amebocyte Lysate (LAL) test was discovered in the 1960s by Levin and Bang.[19] It uses the blood extract of horseshoe crabs, Limulus polyphemus and is mostly known as the indirect animal test. This method can be divided into three basic techniques, the gel-clot, turbidimetric and chromogenic techniques.
Although the LAL test has been accepted as a reliable method for measuring pyrogenicity, it is only specific for the cell wall component of Gram-negative bacteria. Thus, it is not applicable for samples containing a large amount of protein.[11] In the case of endotoxin contamination in veterinary vaccines, the LAL test is not capable for quantifying in vivo endotoxin activities satisfactorily.[12] LAL test is not applicable on samples containing free metal ions as these metal ions can alter the measurement sensitivity, thus reducing the accuracy of the test result. Several types of modification to the method has been employed to reduce the influence of ion. Briefly, the modification involved reconstructing the sample so that the composition of all metal ions in each sample was analogous. A minimum sample dilution (MSD) of 1000-fold was introduced to reduce further the inhibition/enhancement effect of metal ions on the LAL activity without compromising endotoxin sensitivity. 5 mM EDTA was added into each sample to reduce the endotoxin–metal ion interaction. Besides the issues on LAL sensitivity and specificity, the horseshoe crab population is also under threat due to over-fishing for its blood.[13]
Monocyte activation test (MAT)
The Monocyte Activation Test (MAT) has been in existence since 1995. This human blood-based method can quantify all pyrogens which affecting human, and it is a promising alternative to both the RPT and LAL tests [14]. Briefly, either human blood or cell line is directly exposed to the surface of the test material, and the amount of pro-inflammatory cytokine IL-1b is then quantified. This method can detect as little as 10 pg/ml of endotoxin concentration and even lower using cryo-preserved blood.[15] Different types of detection system was created by adopting either human whole blood, cryopreserved blood, peripheral blood mononuclear cells (PBMCs), monocytic cell lines (MONO MAC 6, MM6), or human acute monocytic leukemia cell line (THP-1) as a source for human monocytes, and various read-outs were established.[15] The MAT method is established based on the human fever reaction, and thus most closely reflects the human situation while facing different types of pyrogens.
The Monocyte Activation Test (MAT) has been in existence since 1995. This human blood-based method can quantify all pyrogens which affecting human, and it is a promising alternative to both the RPT and LAL tests [14]. Briefly, either human blood or cell line is directly exposed to the surface of the test material, and the amount of pro-inflammatory cytokine IL-1b is then quantified. This method can detect as little as 10 pg/ml of endotoxin concentration and even lower using cryo-preserved blood.[15] Different types of detection system was created by adopting either human whole blood, cryopreserved blood, peripheral blood mononuclear cells (PBMCs), monocytic cell lines (MONO MAC 6, MM6), or human acute monocytic leukemia cell line (THP-1) as a source for human monocytes, and various read-outs were established.[15] The MAT method is established based on the human fever reaction, and thus most closely reflects the human situation while facing different types of pyrogens.
Recombinant Factor C (rFC) Assay
Recombinant Factor C (rFC) is an endotoxin sensitive recombinant protein. In the assay, the binding of endotoxin activates rFC molecules that cleave a fluorescein substrate, amino-methylcoumarin resulting in the generation of a fluorogenic compound. The fluorescence is measured using 380/440 nm excitation/emission in the absence and presence of endotoxins. The difference in fluorescence is proportional to endotoxin concentrations in the sample. rFC is specific to endotoxin detection eliminating the dependence on nonspecific glycan-binding like that in a LAL assay avoiding false-positive results. The enzymatic sensitivity range to endotoxin is 0.05-500 EU/ml [16].
Recombinant Factor C (rFC) is an endotoxin sensitive recombinant protein. In the assay, the binding of endotoxin activates rFC molecules that cleave a fluorescein substrate, amino-methylcoumarin resulting in the generation of a fluorogenic compound. The fluorescence is measured using 380/440 nm excitation/emission in the absence and presence of endotoxins. The difference in fluorescence is proportional to endotoxin concentrations in the sample. rFC is specific to endotoxin detection eliminating the dependence on nonspecific glycan-binding like that in a LAL assay avoiding false-positive results. The enzymatic sensitivity range to endotoxin is 0.05-500 EU/ml [16].
Biosensor test endotoxin
Following the development of modern biotechnology, electronic technology and microchip technology, a series of bio-electro combined methods has been developed and tested in the past decade. The endotoxin concentration was read directly or indirectly through a sensor and transformed into digital signal and exported to the user interface. Compare to conventional assays, these methods are faster, real-time and labor saving. The biosensor techniques can be divided into three types, electrochemical, optical, and mass-based.
The majority of electrochemical biosensors are based on a principle called Electrochemical Impedance Spectroscopy, or EIS. Performing an EIS requires electrodes to be placed within the solution desired to be tested and delivering a sinusoidal alternating current signal through the solution, usually ranging from 2-10 mV. By varying the frequency of these sinusoidal waves, an impedance spectrum can be created [17]. The electrodes are coated in metal, to reduce electric resistance. Proteins that are highly selective to endotoxin components are bound to these electrodes such that if endotoxins come in contact with the electrode-protein complex, they bind to the proteins.
One of the optical biosensors that have successfully been developed is the endotoxin assay by bioluminescence using mutant firefly luciferase. The biosensing principle is a combination of the LAL reaction and bioluminescence using mutant luciferase (Figure 6) [18]. This method could detect as little as 0.0005 EU/ml endotoxins within 15 min.
Mass-based method is not yet well developed. A method called electromagnetic piezoelectric acoustic sensors or EMPAS that has been touted as being able to measure multiple types of pyrogens, not exclusively endotoxins, as well as being able to detect endotoxins in real-time within human blood plasma [19].
These biosensor-based methods are promising for the need in industry biotechnology, medical devices as well as food and agriculture. However, there is yet product or mature solution can be provided till 2021.
Following the development of modern biotechnology, electronic technology and microchip technology, a series of bio-electro combined methods has been developed and tested in the past decade. The endotoxin concentration was read directly or indirectly through a sensor and transformed into digital signal and exported to the user interface. Compare to conventional assays, these methods are faster, real-time and labor saving. The biosensor techniques can be divided into three types, electrochemical, optical, and mass-based.
The majority of electrochemical biosensors are based on a principle called Electrochemical Impedance Spectroscopy, or EIS. Performing an EIS requires electrodes to be placed within the solution desired to be tested and delivering a sinusoidal alternating current signal through the solution, usually ranging from 2-10 mV. By varying the frequency of these sinusoidal waves, an impedance spectrum can be created [17]. The electrodes are coated in metal, to reduce electric resistance. Proteins that are highly selective to endotoxin components are bound to these electrodes such that if endotoxins come in contact with the electrode-protein complex, they bind to the proteins.
One of the optical biosensors that have successfully been developed is the endotoxin assay by bioluminescence using mutant firefly luciferase. The biosensing principle is a combination of the LAL reaction and bioluminescence using mutant luciferase (Figure 6) [18]. This method could detect as little as 0.0005 EU/ml endotoxins within 15 min.
Mass-based method is not yet well developed. A method called electromagnetic piezoelectric acoustic sensors or EMPAS that has been touted as being able to measure multiple types of pyrogens, not exclusively endotoxins, as well as being able to detect endotoxins in real-time within human blood plasma [19].
These biosensor-based methods are promising for the need in industry biotechnology, medical devices as well as food and agriculture. However, there is yet product or mature solution can be provided till 2021.
In summery, the most commonly used endotoxin test method is still the LAL and rFC methods, since they are both easy to operate, low cost, fast and the reliable result. In US, every drug and medical device that is tested by the US Food and Drug Administration (FDA) must undergo and pass a LAL test [20]. In 2014, the European Directorate for the Quality of Medicines and HealthCare (EDQM) has revised the Guidelines for Using the Test for Bacterial Endotoxin which includes the use of recombinant factor C as a possible alternative method to the LAL test [21]. Rabbit payrogen test is still being used with limitation [22] [23]. Good news is, RPT method is going to be totally replaced soon in the EU [22]. Biosensor methods are still in the concept and without commercial availability. The future is bright but the way remains long (as of the date of this chapter been written, Dec. 12, 2021).
More content? To be continued!
[1] Vaara M, Nikaido H. Outer membrane organization. Handbook of endotoxin. Amsterdam: Elsevier. 1984:1-45.
[2] Rietschel ET, Cavaillon JM. Richard Pfeiffer and Alexandre Besredka: creators of the concept of endotoxin and anti-endotoxin. Microbes and infection. 2003 Dec 1;5(15):1407-14.
[3] Pabst MJ, Johnston Jr RB. Bacterial lipopolysaccharide: a mediator of inflammation. Handbook of inflammation. 1989;6:361.
[4] Rietschel ET, Kirikae T, Schade FU, Mamat U, Schmidt G, Loppnow H, Ulmer AJ, Zähringer U, Seydel U, Di Padova F, Schreier M. Bacterial endotoxin: molecular relationships of structure to activity and function. The FASEB Journal. 1994 Feb;8(2):217-25.
[5] Martich GD, Boujoukos AJ, Suffredini AF. Response of man to endotoxin. Immunobiology. 1993 Apr 1;187(3-5):403-16.
[6] Raetz CR, Ulevitch RI, Wright SD, Sibley CH, Ding A, Nathan CF. Gram‐negative endotoxin: an extraordinary lipid with profound effects on eukaryotic signal transduction 1. The FASEB Journal. 1991 Sep;5(12):2652-60.
[7] Ohno N, Morrison DC. Lipopolysaccharide interaction with lysozyme: Binding of lipopolysaccharide to lysozyme and inhibition of lysozyme enzymatic activity. Journal of Biological Chemistry. 1989 Mar 15;264(8):4434-41.
[8] Anspach FB. Endotoxin removal by affinity sorbents. Journal of biochemical and biophysical methods. 2001 Oct 30;49(1-3):665-81.
[9] Williams KL, editor. Endotoxins: pyrogens, LAL testing and depyrogenation. CRC Press; 2007 Feb 23.
[10] Cooper JF, Levin J, Wagner HN. Quantitative comparison of in vitro and in vivo methods for the detection of endotoxin. The Journal of laboratory and clinical medicine. 1971 Jul 1;78(1):138-48.
[11] Peterbauer A, Eperon S, Jungi TW, Werner ER, Werner-Felmayer G. Interferon-γ-primed monocytoid cell lines: optimizing their use for in vitro detection of bacterial pyrogens. Journal of immunological methods. 2000 Jan 13;233(1-2):67-76.
[12] Usui M, Nagai H, Tamura Y. An in vitro method for evaluating endotoxic activity using prostaglandin E2 induction in bovine peripheral blood. Biologicals. 2013 May 1;41(3):158-61.
[13] Easter MC, editor. Rapid microbiological methods in the pharmaceutical industry. CRC Press; 2003 Mar 19.
[14] Schindler S, Von Aulock S, Daneshian M, Hartung T. Development, validation and applications of the monocyte activation test for pyrogens based on human whole blood. ALTEX-Alternatives to animal experimentation. 2009 Nov 1;26(4):265-77.
[15] Hasiwa N, Daneshian M, Bruegger P, Fennrich S, Hochadel A, Hoffmann S, Rivera-Mariani FE, Rockel C, Schindler S, Spreitzer I, Stoppelkamp S. Evidence for the detection of non-endotoxin pyrogens by the whole blood monocyte activation test. Alternatives to animal experimentation: ALTEX. 2013;30(2):169-208.
[16] Ding JL, Ho B. A new era in pyrogen testing. TRENDS in Biotechnology. 2001 Aug 1;19(8):277-81.
[17] Honeychurch KC. Printed thick-film biosensors. Printed films. 2012 Jan 1:366-409.
[18] Noda K, Goto H, Murakami Y, Ahmed AB, Kuroda A. Endotoxin assay by bioluminescence using mutant firefly luciferase. Analytical biochemistry. 2010 Feb 15;397(2):152-5.
[19] Thompson M, Blaszykowski C, Sheikh S, Romaschin A. A true theranostic approach to medicine: Towards tandem sensor detection and removal of endotoxin in blood. Biosensors and Bioelectronics. 2015 May 15;67:3-10.
[20] Mohanan PV, Banerjee S, Geetha CS. Detection of pyrogenicity on medical grade polymer materials using rabbit pyrogen, LAL and ELISA method. Journal of pharmaceutical and biomedical analysis. 2011 Jul 15;55(5):1170-4.
[21] GMP News: EDQM revises 5.1.10. Guidelines for using the Test for Bacterial Endotoxins [Internet]. [cited 2021 Dec 12]. Available from: https://www.gmp-compliance.org/gmp-news/edqm-revises-5-1-10-guidelines-for-using-the-test-for-bacterial-endotoxins
[22] EDQM Council of Europe, European Pharmacopoeia to put an end to the rabbit pyrogen test [Internet]. [cited 2021 Dec 12]. Available from: https://www.edqm.eu/en/news/european-pharmacopoeia-put-end-rabbit-pyrogen-test
[23] US Food and Drug Administration. Guidance for Industry: Pyrogen and Endotoxins Testing: Questions and Answers [Internet]. [cited 2021 Dec 12]. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-pyrogen-and-endotoxins-testing-questions-and-answers
[24] Weary M, Pearson F. A manufacturer’s guide to depyrogenation. Biopharm. 1988 Apr 29;1(4):22-9.
[25] Ryan J. Endotoxins and cell culture. Corning Life Sciences Technical Bulletin 2004:1–8.
[2] Rietschel ET, Cavaillon JM. Richard Pfeiffer and Alexandre Besredka: creators of the concept of endotoxin and anti-endotoxin. Microbes and infection. 2003 Dec 1;5(15):1407-14.
[3] Pabst MJ, Johnston Jr RB. Bacterial lipopolysaccharide: a mediator of inflammation. Handbook of inflammation. 1989;6:361.
[4] Rietschel ET, Kirikae T, Schade FU, Mamat U, Schmidt G, Loppnow H, Ulmer AJ, Zähringer U, Seydel U, Di Padova F, Schreier M. Bacterial endotoxin: molecular relationships of structure to activity and function. The FASEB Journal. 1994 Feb;8(2):217-25.
[5] Martich GD, Boujoukos AJ, Suffredini AF. Response of man to endotoxin. Immunobiology. 1993 Apr 1;187(3-5):403-16.
[6] Raetz CR, Ulevitch RI, Wright SD, Sibley CH, Ding A, Nathan CF. Gram‐negative endotoxin: an extraordinary lipid with profound effects on eukaryotic signal transduction 1. The FASEB Journal. 1991 Sep;5(12):2652-60.
[7] Ohno N, Morrison DC. Lipopolysaccharide interaction with lysozyme: Binding of lipopolysaccharide to lysozyme and inhibition of lysozyme enzymatic activity. Journal of Biological Chemistry. 1989 Mar 15;264(8):4434-41.
[8] Anspach FB. Endotoxin removal by affinity sorbents. Journal of biochemical and biophysical methods. 2001 Oct 30;49(1-3):665-81.
[9] Williams KL, editor. Endotoxins: pyrogens, LAL testing and depyrogenation. CRC Press; 2007 Feb 23.
[10] Cooper JF, Levin J, Wagner HN. Quantitative comparison of in vitro and in vivo methods for the detection of endotoxin. The Journal of laboratory and clinical medicine. 1971 Jul 1;78(1):138-48.
[11] Peterbauer A, Eperon S, Jungi TW, Werner ER, Werner-Felmayer G. Interferon-γ-primed monocytoid cell lines: optimizing their use for in vitro detection of bacterial pyrogens. Journal of immunological methods. 2000 Jan 13;233(1-2):67-76.
[12] Usui M, Nagai H, Tamura Y. An in vitro method for evaluating endotoxic activity using prostaglandin E2 induction in bovine peripheral blood. Biologicals. 2013 May 1;41(3):158-61.
[13] Easter MC, editor. Rapid microbiological methods in the pharmaceutical industry. CRC Press; 2003 Mar 19.
[14] Schindler S, Von Aulock S, Daneshian M, Hartung T. Development, validation and applications of the monocyte activation test for pyrogens based on human whole blood. ALTEX-Alternatives to animal experimentation. 2009 Nov 1;26(4):265-77.
[15] Hasiwa N, Daneshian M, Bruegger P, Fennrich S, Hochadel A, Hoffmann S, Rivera-Mariani FE, Rockel C, Schindler S, Spreitzer I, Stoppelkamp S. Evidence for the detection of non-endotoxin pyrogens by the whole blood monocyte activation test. Alternatives to animal experimentation: ALTEX. 2013;30(2):169-208.
[16] Ding JL, Ho B. A new era in pyrogen testing. TRENDS in Biotechnology. 2001 Aug 1;19(8):277-81.
[17] Honeychurch KC. Printed thick-film biosensors. Printed films. 2012 Jan 1:366-409.
[18] Noda K, Goto H, Murakami Y, Ahmed AB, Kuroda A. Endotoxin assay by bioluminescence using mutant firefly luciferase. Analytical biochemistry. 2010 Feb 15;397(2):152-5.
[19] Thompson M, Blaszykowski C, Sheikh S, Romaschin A. A true theranostic approach to medicine: Towards tandem sensor detection and removal of endotoxin in blood. Biosensors and Bioelectronics. 2015 May 15;67:3-10.
[20] Mohanan PV, Banerjee S, Geetha CS. Detection of pyrogenicity on medical grade polymer materials using rabbit pyrogen, LAL and ELISA method. Journal of pharmaceutical and biomedical analysis. 2011 Jul 15;55(5):1170-4.
[21] GMP News: EDQM revises 5.1.10. Guidelines for using the Test for Bacterial Endotoxins [Internet]. [cited 2021 Dec 12]. Available from: https://www.gmp-compliance.org/gmp-news/edqm-revises-5-1-10-guidelines-for-using-the-test-for-bacterial-endotoxins
[22] EDQM Council of Europe, European Pharmacopoeia to put an end to the rabbit pyrogen test [Internet]. [cited 2021 Dec 12]. Available from: https://www.edqm.eu/en/news/european-pharmacopoeia-put-end-rabbit-pyrogen-test
[23] US Food and Drug Administration. Guidance for Industry: Pyrogen and Endotoxins Testing: Questions and Answers [Internet]. [cited 2021 Dec 12]. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-pyrogen-and-endotoxins-testing-questions-and-answers
[24] Weary M, Pearson F. A manufacturer’s guide to depyrogenation. Biopharm. 1988 Apr 29;1(4):22-9.
[25] Ryan J. Endotoxins and cell culture. Corning Life Sciences Technical Bulletin 2004:1–8.